Take Action: Ask congress to help regulators take down websites that sell drugs illegally
The “Domain Reform for Unlawful Drug Sellers” or “DRUGS” Act would require domain name sellers to lock and suspend websites when regulators or law enforcement show evidence that they are selling drugs illegally.
What you can do
We can help you speak to your Senators to endorse this legislation. Use this form to contact your legislators, or reach out to us at editors@safemedicines.org to craft an individual letter.
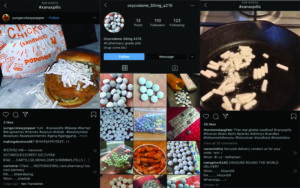
Photos from websites selling drugs illegally
Synopsis:
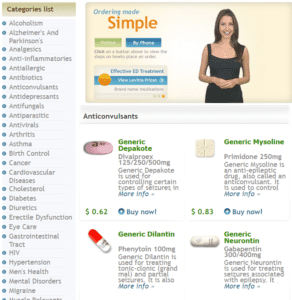
95 percent of websites selling prescription drugs are operating illegally. Many sell fake medicine or fill orders for controlled substances without prescriptions, and with no legal way to force domain sellers to shut down sites that host criminal activity, it’s hard to stop them.
The “Domain Reform for Unlawful Drug Sellers” or “DRUGS” Act would change that by requiring domain name sellers to lock and suspend websites when regulators or law enforcement show evidence that they are selling drugs illegally.
This would be a game changer for patient safety
- The DRUGS Act could have locked the fake pharmacy website that sold painkillers made with fentanyl analogues BEFORE one of its customers died in 2017.
- Canadian pharmacist Andrew Strempler could not have sold Americans fake Lipitor and cancer medicine in 2006 without his RxNorth website.
- And it could put the brakes on any number of scam websites that have popped up selling counterfeit vaccines and treatments for COVID-19.
The DRUGS act could be a critical tool to stop criminals from selling fentanyl and counterfeit medicines to Americans online. We support it, and we hope you will, too.
Current Status:
The “Domain Reform for Unlawful Drug Sellers” or “DRUGS” Act was introduced in both the Senate (S. 3399) and the House (H.R. 6352) in December 2021. The bills were referred to the Senate Committee on Health, Education, Labor, and Pensions and the House Committee on Energy and Commerce.
Background / resources
- Factsheet on websites selling opioids, fentanyl, and other controlled substances (PSM)
- Disrupting Illegal Online Pharmacies: Lock-and-Suspend as a Tool to Protect Patients (NABP)
- Factsheet on the DRUGS Act: Text version / Graphical version (ASOP)
- S.3399: The Domain Reform for Unlawful Drug Sellers Act
The Details
The bill would amend the Federal Food, Drug, and Cosmetic Act to establish a process to lock and suspend web domain names used to sell drugs illegally.
Reporting and response: Registry operators would be required to lock a domain name less than 24 hours after being notified by a trusted notifier that it’s being used to sell drugs and suspend it within seven days of notification.
Trusted notifiers include: the Food and Drug Administration (FDA), the Department of Justice , the Drug Enforcement Administration, state attorneys general, state boards of pharmacy, nonprofits with the membership of these officials, contractors for the FDA and other entities FDA recognizes as valid.
Appeal: The person or company to whom the domain name is registered can appeal by contacting the registry to request the source and email address of the notifier and providing a copy of pharmacy or appropriate medical licenses for each place sales are occurring. If the appeal is successful, the registry should release the domain within 15 days.
Videos about lock and suspend

Locking and suspending domain names can disrupt fake pharmacy websites—and the NABP agrees.

Learn about websites selling Narcotics and how the DRUGS Act could stop this. Also see our factsheet.

Learn about websites illegally dispensing dangerous HIV and HEPC medicine and how the DRUGS Act could stop this.

PSM Coverage of Fake Online Pharmacies
See older news on PSM's pinboard.