Fake HIV medication has made its way to U.S. pharmacies—and patients
Since 2019, federal prosecutors, drug manufacturers, and patient safety organizations have uncovered extensive criminal schemes targeting HIV medicines at every point in the supply chain. These schemes undermine trust in the healthcare system, and they endanger patients who rely on high quality, effective medicine to control viral loads and prevent HIV transmission.
Where are these supply chain breaks coming from?
- Wholesalers
- Pharmacies
- Insurers
- Pharmacy-to-pharmacy marketplaces
- Buy back schemes on social apps
Wholesalers sold licensed pharmacies secondhand medicine with fake pedigrees
In December 2020 and August 2021, Janssen Pharmaceuticals and Gilead Sciences issued the alarming news that counterfeit HIV medicines were circulating in the United States.
Then, in January 2022, the Wall Street Journal broke an astonishing story: Gilead Sciences was suing a ring of drug sellers and distributors that allegedly sold 85,247 of the counterfeit bottles worth more than $250 million to U.S. pharmacies.
Gilead Sciences v Safe Chain Solutions, et al.
The suit accused a complex network of 140 companies and individuals across 13 states of selling fake and diverted medications to U.S. pharmacies using forged track and trace documents.
The network collected bottles of medicine from patients, stripped them of identifying information, refilled and resealed them, and laundered them through distributors with forged pedigrees so that pharmacies didn't realize there was anything wrong with them.
Patients received closed containers of vital medicine that looked legitimate, but sometimes the bottles contained the wrong HIV treatment, over-the-counter painkillers, or actual pebbles. In one case, a patient's sealed bottle contained an antipsychotic medicine that rendered them unable to walk or speak.
Gilead also filed Gilead Sciences v Peter Khaim, which identified a second network of individuals and companies alleged to be engaged in the same activity with New York and New Jersey pharmacies. Some of these defendants used pharmacy-to-pharmacy marketplaces to move their diverted medicine.
Over several years, Gilead settled charges with hundreds of defendants, including SafeChain Solutions, which agreed to pay Gilead $2.7 million in February 2024.
Gilead Sciences' amended complaint (September 2022) describes the scheme.
Criminal cases related to Gilead's civil suits
USA v Lazaro Hernandez: Lazaro Roberto Hernandez, who Gilead characterized as a kingpin of the black market ring, received a 15-year federal prison sentence and a forfeiture of $238.8 million after admitting to “running a criminal enterprise” and diverting medicines.
USA v Armando Herrera: Another “kingpin,” Armando Herrera, received a 51-month sentence for introducing adulterated and misbranded drugs into interstate commerce.
USA v Adam Brosius, Patrick Boyd and Charles Boyd: The owners of Maryland distributor SafeChain Solutions, Adam Brosius, Patrick Boyd and Charles Boyd were charged with distributing misbranded and adulterated drugs, trafficking medicine products with false documentation, and wire fraud in 2024.
Brosius pleaded guilty to wire fraud in April 2025 and received an eight-year sentence in October 2025.
The Boyd brothers were convicted by jury of conspiracy to introduce adulterated and misbranded drugs to defraud the United States, conspiracy to traffic in medical products with false documentation, and conspiracy to commit wire fraud in October 2025, and were sentenced to decades in prison in March 2026.
See trial exhibits and PSM coverage of this case here.
USA v Steven Diamantstein: Scripts Wholesale owner Steven Diamantstein was indicted in New Jersey federal court in 2023 for allegedly acquiring unapproved, diverted prescription medicines and fraudulently selling to pharmacies with falsified pedigrees and labels to make them look like legitimate drugs. This case is still in progress.
USA v Boris Aminov: Christy Corvalan, the owner of a Bronx, New York, pharmacy named in Gilead Sciences v Safe Chain was criminally charged along with nine other defendants in USA v Boris Aminov for trafficking in black-market HIV medication and defrauding Medicaid, Medicare, and private insurance companies out of at least $20 million. As of December 2025, the Aminov defendants had been sentenced to a cumulative 39 years in prison, with fines and forfeitures of more than $55 million.
On-app buyback schemes
Medicine buyback operations, which sometimes claim to be "Buyers Clubs" making medicines available to people who cannot afford them, are the first component of counterfeit schemes like the one Gilead v Safe Chain exposed.
"Collectors" post to apps like Grindr to find patients they can buy medicine, and sometimes empty bottles, from and sell them to distributors that eventually funnel them to unsuspecting pharmacies. They pose as people performing a public service, but they are the first step in a criminal chain that hurts patients.
PSM and ADAP Advocacy issued an alert and a flyer about this issue in 2024.
In progress: A New York City pharmacy allegedly sold counterfeit Biktarvy
Gilead Sciences v Heal the World Pharmacy
A complaint filed by Gilead Sciences in March 2025 asks for an injunction to stop Queens, New York-based Heal The World Pharmacy from selling its products because they are allegedly selling counterfeits. The filing says that a patient called Gilead in January because their bottle of Biktarvy—which comes to patients sealed—was short eight tablets. Gilead investigators examined the bottle. It had an old lot number, a fake expiration date, and typographical errors that marked it as counterfeit.
Images from the counterfeit bottle shared in Gilead's complaint:

"Only" should not be capitalized and "Manfacturaded" is a typo.

Court filings
- March 17, 2025
- March 17, 2025
- March 17, 2025
- April 7, 2025
- April 9, 2025
- April 14, 2025
- April 14, 2025
- April 14, 2025
- April 14, 2025
- April 18, 2025
- April 17, 2025
- April 18, 2025
- April 28, 2025
- May 1, 2025
- June 12, 2025
- July 31, 2025
- August 7, 2025
- September 5, 2025
- September 5, 2025
- September 5, 2025
- September 5, 2025
- November 21, 2025
- November 21, 2025
- November 21, 2025
- January 18, 2026
- January 19, 2026
- January 19, 2026
- January 18, 2026
- February 17, 2026
- February 17, 2026
PSM coverage

Watch PSM's May 2025 video about this case.
Insurers dodging HIV med prices with unregulated, foreign medicine
Recently, employers who sponsor self-funded insurance plans are steering patients into filling high-cost medications from “alternative” sources, including importing them from foreign pharmacies. “Alternative Funding Program” vendors help health plans facilitate these efforts.
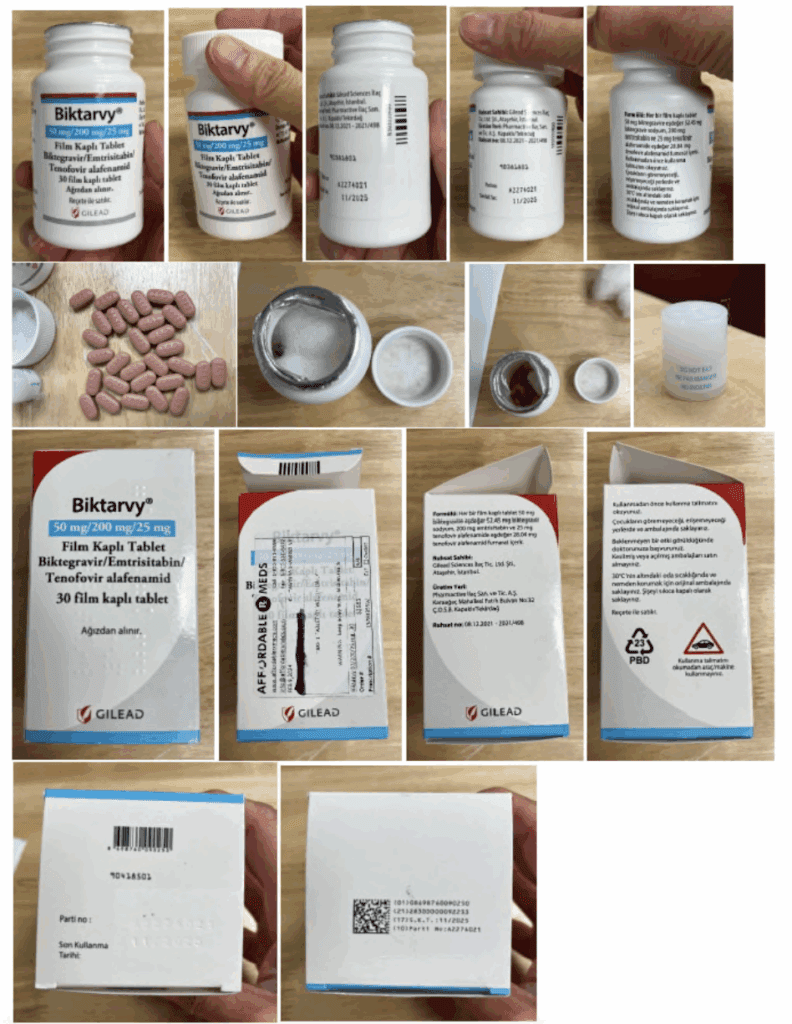
In 2024, a HIV patient in Maryland received illegally imported Turkish Biktarvy with no English labeling through a network of companies working with his insurance. His prescription had been refused at his local pharmacies, and shunted without his knowlege to a foriegn supplier. A lawsuit over this practice is now setting significant legal precedents.
Gilead Sciences v Meritain Health, et al
Gilead Sciences filed a complaint in December 2024 asking for a preliminary injunction to stop a network of companies they alleged were supplying patients with non-FDA approved, imported versions of Gilead's medicines via alternative funding programs (AFPs) to save their employer-sponsored health plans money. Gilead learned about the importation scheme after a patient who filled his HIV prescription with his health insurance received medicines mailed from a retail pharmacy in Turkey. The initial defendants in this case were:
- Meritain Health, a health benefits administrator;
- ProAct Inc., a pharmacy benefit manager;
- Gregory Santulli, the owner of Rx Valet, an AFP charged with finding the least expensive sources of medicine, and Advanced Pharmacy, a licensed U.S. pharmacy;
- Affordable Rx Meds, a broker that contracts with foreign pharmacies; and
- Fetih Eczanesi, a Turkish pharmacy.
In June 2025, a federal judge granted Gilead's request for a temporary injunction in an extensive opinion affirming that international sourcing of drugs through AFPs was not legal, and that companies and people facilitating this activity might be liable for it.
In September 2025 Gilead submitted an amended complaint naming CanaRx, ElectRx, ScriptSourcing, and several of their executives as defendants, alleging that they, too, had acted as alternative funding programs for Meritain Health and ProAct. Courts granted Gilead an injunction for these defendants in October 2025.
Court filings
December 2024:
- Complaint, Gilead Sciences v Meritain Health et al
June 2025:
- Memorandum opinion, Gilead Sciences v Meritain Health et al
September 2025:
- First amended complaint, Gilead Sciences v Meritain Health et al
- Memorandum of law in support of plaintiffs’ motion fortemporary restraining order, expedited discovery order, and order to show cause for a preliminary injunction, Gilead Sciences v Meritain Health et al
PSM coverage on AFPs

Watch PSM's video about this case.
More about counterfeit HIV/AIDS treatments
PSM handouts
Additional PSM coverage:
See older news about counterfeit and diverted HIV drugs on our pinboard.