Search:
Filter:
Sort:
Is personal drug importation legal in the context of alternative funding programs? The FDA says no.
Are self-funded health plans allowed to import medicine because of “Section 804” or “Florida’s importation plan”? Also, no.
United States District Court District of Utah USA v Justin Bradley Watkins Indictment Filed April 1, 2026 Read the document.
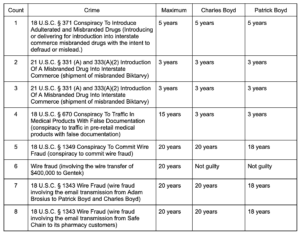
Adam Brosius and Charles and Patrick Boyd were proprietors of Safe Chain Solutions, a Maryland drug wholesaler that sold distributed misbranded and adulterated drugs and trafficked medicine products with false documentation. The men have been sentenced to a combined 46 years in prison.
The doctor allegedly bought the unapproved drugs through a middleman and relabeled them before selling them to patients.
CVS Health’s Caremark deal mirrors the deal the FTC struck with Express Scripts last month, a person familiar with the case told Reuters.
A federal judge in Alaska sentenced rheumatologist Dr. Claribel Tan to six-and-a-half years in prison for administering substandard drugs to patients while fraudulently billing insurers
Online pharmacy-to-pharmacy marketplaces help with inventory management, but they also require vigilance because they carry a higher risk of being exploited by criminals selling black market medicines.
Wire fraud charges drove much higher penalties than the shipping of unsafe medicine into the supply chain, and the Boyds’ sentences were based on the government proving high-dollar, deliberate fraud.
United States District Court Southern District of Texas, Houston Division USA v Sanjay Kumar Judgment Filed March 2026 Read the document.