The FDA speaks up about AFPs and drug importation
April 8, 2026
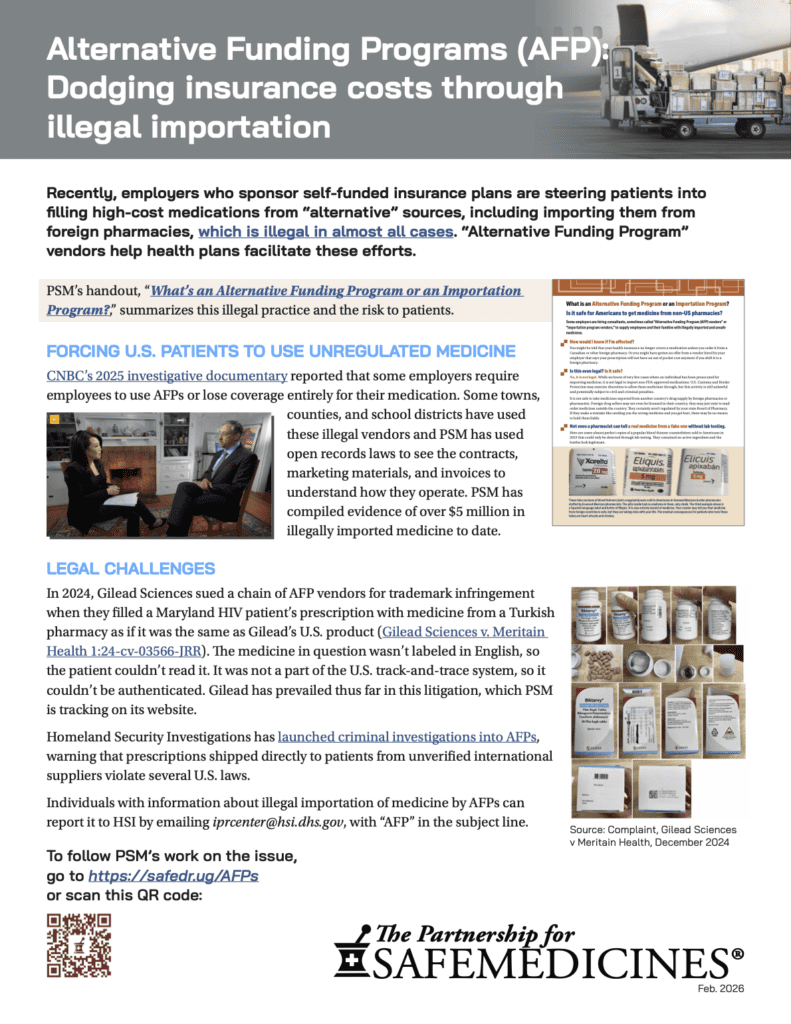
Is personal drug importation legal in the context of alternative funding programs? The FDA says no.
Are self-funded health plans allowed to import medicine because of “Section 804” or “Florida’s importation plan”? Also, no.
In March 2024, patient advocacy group Aimed Alliance filed a citizen petition asking the U.S. Food and Drug Administration to issue guidance clarifying whether alternative funding programs (AFPs) were legally permitted to use drug importation programs to lower insurance costs for employer-sponsored health plans. This guidance was necessary, Aimed Alliance argued, because AFPs were building international importation into self-funded health plans, essentially forcing employees to use medicines made outside FDA regulation to their own detriment, and in violation of the Food, Drug, and Cosmetic Act. AFP vendors maintain that the FDCA’s personal importation policy allows this. AIMED Alliance asked the agency to clarify this.
And they did. The agency said it wasn’t necessary to say anything because they’ve already said it’s illegal and its position is clear. The agency’s response plainly stated that:
- Drug importation is only permissible within authorized Section 804 importation programs. Regardless of what your AFP vendor tells you, if your name isn’t Ron DeSantis, you don’t qualify for a Section 804 program importation exception to import Canadian medicine.
- It is illegal in most cases for individuals to import drugs into the United States for personal use.
- “Many” drugs purporting to be Canadian sold to Americans “are obtained from ever-evolving illicit sources.”
The agency also emphasized that the cost of a drug is not a factor in its case-by-case decisions around personal importation and reinforced the need for clear reporting pathways for potentially unauthorized practices.
The agency declined to conduct more enforcement, but that isn’t surprising as enforcement isn’t typically started because of a citizen’s petition. They also could not have told us about existing, ongoing investigations of AFP vendors.
Bottom line:
If you’re an employer with a self-employed health plan that has a vendor pitching you international medicine fulfillment and telling you it’s legal because of “Section 804,” “Florida’s Canadian drug importation plan,” or “personal importation,” the FDA has explained that this is all rubbish. None of this is legal. You’ll want to get a good legal indemnity clause from your vendor in case they are the next enforcement example by the FDA.
You’ll also want to consider whether you need to be indemnified civilly, since Gilead Sciences, Novartis, and Genentech have all filed civil trademark violation suits against companies involved in this trade. Trademark violations (via the Lanham Act) are judged on strict liability. Your vendor’s promises of compliance will not be a legal defense against your liability for trademark infringement.