Prescription drug freight fraud report, January 2026
In November and December 2025, thousands of shipments of drugs that have been popularized by social media were imported into the United States, along with rivaroxaban (Xarelto) and antibiotics. They were imported into the country with a wide range of product code classifications, some appropriate, and others far less so. Most shipments were declared as coming from reputable manufacturing facilities, but others declared manufacturing addresses that are blatantly unbelievable.
This surge is happening against a clear backdrop: the social media popularization of drugs that should only be obtained with a prescription from the legitimate, legal supply chain. Platforms are flooding users with influencer testimonials, before-and-after photos, and ads promising “life-changing weight loss medications” as “affordable,” “doctor-trusted,” and “formulated in the USA.” Everything from weight loss and hair loss treatments to acne medications feels like it's available at the click of a button.
Read more:
Knockoff weight loss drugs from illegal foreign sources (PSM report, March 2025)
Review previous updates on our pharmaceutical border security page
To understand what was entering the country, we examined import records for these products using the FDA’s Import Entry Data Search and cross-referenced shipment data against multiple FDA systems. We checked declared manufacturers on the manifests to see if they were FDA-registered facilities, reviewed container-level manifests, analyzed product code classifications, and checked outcomes of shipments.
We identified dozens of shipments tied to unregistered facilities, including shipments measured in kilograms and shipments explicitly labeled as active pharmaceutical ingredients (APIs). But the most important story wasn’t just where these shipments said they came from, but how they were described to the FDA.
Product codes set the tone
Before a drug is imported to the US, enforcement decisions begin with paperwork. Importers must declare a product code indicating the industry, class, process in which it’s made, and product in the shipment.
Drug product codes are detailed and include a Process Indicator Code (PIC), which is the part of the product code that signals to an FDA inspector if a drug is an active pharmaceutical ingredient. Cosmetics are different.
Cosmetic product codes do not have a PIC. The FDA defines cosmetics as products that are intended to be applied to the body, “for cleansing, beautifying, promoting attractiveness, or altering the appearance." In addition to the treatment of a condition, the FDA defines drugs as products that are “intended to affect the structure or any function of the body of man or other animals."
Cosmetic product codes are designed for relatively low-risk lotions, liquids, and raw materials. When a shipment enters under a cosmetic code, it moves through a framework that was never intended for pharmacologically active substances. One shipment we saw made that distinction impossible to ignore.
Is it a cosmetic or a pharmaceutical ingredient? It’s a new place to hide.
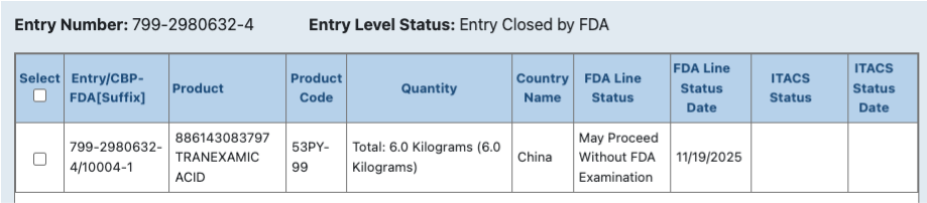
The shipment in question was imported under a product code that is listed as a general “cosmetic raw ingredient,” but the ingredient listed in ITACS, 6 kilograms of tranexamic acid, is the initial red flag. The ingredient exists in a classic “cosmeceutical” grey area, a drug that could be used as both a pharmaceutical or a cosmetic product. At that point, it comes down to the intended use of the product.
When researching the product, it becomes clear that the manufacturer, Spec-Chem Industry Inc., which is an unregistered facility, intends for the product to be used to permanently alter the body, something the FDA says qualifies an ingredient as a drug. According to their website, the product “can remove pigmentation, whitening, and lighten spots, such as pigmentation after sun exposure, dark spots (chloasma, freckles, senile lentils [solar lentigines — ED], etc.), sensitive skin, acne and inflammation, post-operative care after laser, pulse light treatment, etc.”
An ocean of shipments come into the U.S. coded as cosmetics. We have not conducted a systematic review of these yet.
Miscoding medicines that may be very serious as simple antibiotics
Throughout 2025, we’ve identified hundreds of shipments with product codes claiming to be antibiotics with an ITACS description listing something else entirely. In November and December, these mismatches included cough drops, blood-pressure medications, beta blockers, kidney cancer drugs, and even a hepatitis B vaccine.
In total, 76 of the 437 (about 17%) of the antibiotic shipments we studied showed classification mismatches when reviewed at the manifest level.
The shipments below were incorrectly coded as antibiotics.
| Shipment ID | Was the manufacturer registered? | Product code | ITACS declaration |
| SCS-5655213-2/10002/1 | Unregistered | 56YYR99 | Semax 1%, 60 MG: a synthetic nootropic peptide, used to enhance cognitive function, neuroprotection, and stroke recovery. |
| AEK-9113853-5/50003/1 | Unregistered | 56YDY99 | Metoprolol: a beta-blocker used for heart conditions like high blood pressure, chest pain, and heart failure. |
| 799-5107043-5/10003/1 | Unregistered | 56YCY99 | Doxylamine succinatom pyridoxine HCL: The combination of doxylamine and pyridoxine is used to treat nausea and vomiting in pregnant women |
| AEK-9113853-5/60003/1 | Unregistered | 56YDY99 | Cilacar: a calcium channel blocker medication used to treat high blood pressure (hypertension) and prevent angina. It works by relaxing blood vessels to improve blood flow, reducing the risk of heart attacks and strokes. |
| 231-7001807-8/10003/1 | Registered | 56YCP99 | Torisel: a cancer medicine that interferes with the growth and spread of cancer cells in the body. Torisel is used to treat cancer of the kidneys, also called renal cell carcinoma. |
| AEK-9113853-5/100003/1 | Registered | 56YDY99 | Oxetol hr 600 mg tablets: anticonvulsant (anti-epileptic) medication containing Oxcarbazepine, used to control partial-onset seizures in adults and children (6+ years). |
| SCS-6481683-3/130002/1 | Registered | 56YBO99 | IMMUNOHBs is a human hepatitis B immunoglobulin solution used for passive immunization to prevent hepatitis B virus (HBV) infection. It provides immediate, short-term protection (3–6 months) and is used for post-exposure prophylaxis, especially in infants of HBsAg-positive mothers or following accidental exposure. |
While this could be something as benign as a paperwork mistake on behalf of the manufacturer, we can’t help but wonder if bad actors are intentionally misrepresenting the risks of the product they are trying to ship into the US to avoid scrutiny. If so, they are using the historic strain on the FDA against the agency to subvert its mission to protect Americans.
Border security in the age of viral medicine
The rise in social media attention around specific drugs has closely tracked increased cross-border movement of those same products. Weight-loss drugs such as semaglutide and tirzepatide are widely discussed online and often marketed as accessible, affordable, and easy to obtain. Telehealth platforms and med spas amplify that demand, promising convenience and rapid delivery.
That demand shows up at the border in multiple forms, some shipments labeled as active pharmaceutical ingredients, and others declared as finished drug products in small quantities. Both categories reveal supply-chain vulnerabilities. In November and December, we analyzed:
- 243 shipments of semaglutide, including 857.11 kilograms labeled as API, and 13 more shipments labeled as API for compounding, with no weight given.
- 528 shipments of tirzepatide, with 37 (7%) tied to unregistered facilities.
- 158 shipments of minoxidil, 73 (46%) associated with unregistered manufacturers.
- 111 shipments of spironolactone, including 13 kilogram-scale imports totalling 21,176 kilograms of the drug.
Alongside API shipments were finished-product imports that raised separate but related concerns.
One suspicious semaglutide shipment came from a Canadian pharmacy that illegally advertises “providing Americans and people around the world with access to medication at the same prices most Canadians pay.” This small quantity of semaglutide got into the country, but the FDA stopped another attempted shipment from pharmacy.cloud, a Canadian online pharmacy in November 2025.
Another suspect shipment listed its manufacturer as a Marriott hotel connected to Montreal Airport. While this was a small import of tirzepatide, it was let into the country. A one kilogram shipment of tirzepatide from Seenergie Inc. was let into the country despite it being an unregistered facility.
In every release of our freight fraud report, we’ve identified residential addresses that claim to be foreign manufacturers importing small shipments of weight loss medications. One notable shipment, claiming to be manufactured by Novo Nordisk with an address in a Toronto suburb, imported one piece of semaglutide. There were three additional semaglutide, 11 tirzepatide, eight Minoxidil, two spironolactone, and two Xarelto shipments claiming to be manufactured at residential addresses.
Minoxidil, often framed online as cosmetic or wellness-related, had the highest proportion of unregistered facilities. In one case, a Colombian hair-transplant clinic imported one kilogram of minoxidil. In another, shipments of “Minoxytop” arrived from an overseas manufacturer advertising itself as FDA-approved, despite having no registered facilities on file. Even retail destinations appeared in the data, including a home-goods store in Tennessee listed as the foreign manufacturer on a shipment entering the U.S.
These examples are not evidence of compounded drugs crossing the border, but they are evidence of a fragmented and inconsistent supply chain feeding intense market demand. Whether shipments are labeled as APIs or as finished products, the recurring themes are the same: unregistered facilities, implausible manufacturing sites, and successful entry despite red flags. All medicine made in the U.S., whether it's commercially manufactured or compounded, depends on the integrity of the upstream supply chain, particularly the quality and traceability of active ingredients.
Enforcement is working, but more is needed
None of this suggests a lack of enforcement. Refusal data shows that Customs and Border Protection and FDA staff are actively identifying and stopping problematic shipments. Officers routinely flag misbranding, unapproved drugs, and illegitimate facilities, even when shipments are small or disguised.
About 22% of rivaroxaban (Xarelto) shipments from illegitimate manufacturers were refused for exactly these reasons. For semaglutide, about 20% of shipments from illegitimate manufacturers were refused.
Still, the scales of social-media-driven demand and compounding, and the prevalence of classification ambiguities, demonstrate the need for better upstream visibility. It also underscores the need to bolster the FDA’s resources; in its current strained capacity, it is impossible for the agency to screen every suspect shipment. Each mismatch or misclassified shipment highlights a point of potential patient risk that could be addressed before a product reaches a pharmacy or med spa. The FDA needs additional resources to fulfill its lifesaving mission of protecting Americans.
For patients, the consequences are real. Misclassified or misdescribed raw ingredients increase the likelihood that compounded medications are inconsistent, contaminated, or otherwise unsafe before they ever reach the syringe. Social-media hype and online convenience amplify those risks by accelerating demand and encouraging sourcing shortcuts.
Access to our data
PSM makes our shipment-level data available to a group of manufacturer brand protection teams, law enforcement, and regulators.
Our policy recommendations
Short term:
- Shipments with an invalid FEI number should be refused or detained without physical examination.
Medium term:
- FDA should be given the authority to mandate the destruction of violative shipments whose manifest states they were made in non-FDA-registered facilities. Shippers should pay the cost of destruction and present proof of destruction to the FDA.
- FDA and CBP should refuse or detain shipments of medical products whose manifest claims that they were manufactured at a site that is not a registered FDA facility.
Long term:
- CBP should be allowed to process, refuse, detain, and mandate the destruction of shipments that cannot be released because they violate the Food, Drug, and Cosmetic Act. Currently, they can only act on trademark violations and controlled substance violations (through an MOU with the Drug Enforcement Administration). Adding their manpower to the FDA’s will keep Americans safer.