Posts Tagged ‘FDA Alert’
FDA Alert: Florida Company’s Dietary Supplement Recalled for Containing Prescription Drug Ingredients
STOP CLOPEZ CORP is voluntarily recalling one lot of Schwinnng capsules to the consumer level. FDA analysis has found the Schwinnng products to contain Nortadalafil. Nortadalafil is an active drug ingredient known for the treatment of male erectile dysfunction. The presence of Nortadalafil in Schwinnng capsules makes it an unapproved new drug for which the safety and efficacy have not been established and, therefore subject to recall.
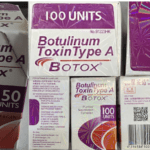
[...]FDA alert: Counterfeit version of Botox found in multiple states
The products appear to have been purchased from unlicensed sources. Medications purchased from unlicensed sources may be misbranded, adulterated, counterfeit, contaminated, improperly stored and transported, ineffective and/or unsafe.
[...]FDA warns consumers not to use counterfeit Ozempic (semaglutide) found in U.S. drug supply chain
FDA continues to investigate counterfeit Ozempic (semaglutide) injection 1 milligram (mg) in the legitimate U.S. drug supply chain and has seized thousands of units of the product. FDA is aware of five adverse events from this lot.
[...]FDA Alert: Ginseng Compound Recalled for Containing Undeclared Drug Ingredients
This is a reprint of an FDA Alert. 8th Avenue Pharmacy Issues Voluntary Nationwide Recall of Notoginseng Formula Special Gout Granule Due to the Presence of Hidden Drug Ingredients, Diclofenac and Dexamethasone When a company announces a recall, market withdrawal, or safety alert, the FDA posts the company’s announcement as a public service. FDA does…
[...]FDA Alert: Pain Relief Dietary Supplements Recalled Due to Presence of Diclofenac
Risk Statement: Consumption of undeclared diclofenac could result in serious adverse events that include cardiovascular, gastrointestinal, renal, and anaphylaxis in patients taking concomitant NSAIDs and/or anticoagulants, such as Warfarin, in those who have allergies to diclofenac, or those with underlying cardiovascular, gastrointestinal, renal, and hepatic illnesses.
[...]FDA Alert: Suprimo Imports Warned by FDA After Reports of Illness and Death Due to Prescription Drug Ingredients in Their Dietary Supplements.
“On April 20, 2022, FDA warned consumers not to purchase or use “Artri Ajo King” as FDA has received adverse event reports, including of liver toxicity and death, associated with the use of “Artri Ajo King” and similarly named products. While the Agency has not sampled and tested this product from your inventory to date, be advised of our serious concern about the safety of these products and that it is your legal responsibility under federal law to ensure this product does not contain any undeclared and potentially harmful ingredients.”
[...]FDA Alert: Three Dietary Supplement Consumer Warnings, All Contain Prescription APIs
The FDA has issued consumer warnings on three different dietary supplement sold as herbal compounds intended to treat joint pain, rheumatoid arthritis, gout, and liver detoxification. Each product contains undeclared prescription drug ingredients.
[...]FDA Alert: Dietary Supplement Recalled Due to Presence of Prescription Drug Ingredients
FOR IMMEDIATE RELEASE – April 26, 2023– West Sacramento, CA, Gear Isle is voluntarily recalling the following products listed in the table below to the consumer level. FDA analysis has found the products to be tainted with prescription drug ingredients.
[...]FDA Alert: Dietary Supplement For Vision Care Recalled Due to Unapproved and Dangerous Ingredients
TruVision Health LLC is recalling the dietary supplement products listed below because they contain the unapproved dietary ingredients hordenine and/or octodrine/DMHA (1,5-Dimethylhexylamine).
[...]FDA Alert: Nano Silver Dietary Supplement Triggers Recall
This recall has been initiated because the Product is labeled as a dietary supplement that, in the opinion of the government, makes unsubstantiated health claims that the product will prevent, treat, or cure COVID-19.
[...]