Drug Importation in Texas: An Overview
Current status:
The Texas Legislature introduced House Bill 25, the Wholesale Prescription Drug Importation Act, in November 2022. PSM shared an analysis of the bill with the Texas House committee that you can read.
The bill which has been signed by the Governor, will require the executive commissioner of Texas's Health and Human Services Commission to take action to establish Canadian drug importation beginning in September 2023. The agency shared a report about the program in December 2023.
Synopsis / Media Contact
H.B. 25 requires Texas’ Health and Human Services Commission to design a program for bulk importing prescription medicines under 21 USC 384 of the U.S. Food, Drug, and Cosmetics Act, more commonly known as a Section 804 State Importation Program (SIP).
For over two decades these programs have proven to be impossible to build safely because:
- Canada refuses to release bulk medications to these programs and has taken steps to restrict U.S. access to their drug supply;
- Canada does not have a Track and Trace program;
- The cost of testing imported medication far outweighs the savings; and
- Medicaid programs already get medicine cheaper than Canadian provinces, reducing any potential savings.
This legislation has been pushed without concern for the consequences. As many states discovered during the pandemic trying to buy hard-to-find N95 masks, buying medical products that aren't readily available creates great risk of counterfeit products. PSM believes that the Texas legislature is forcing it's patients and pharmacists to take unacceptable risks with patient health and pharmacist liability by enacting this program without looking at the risks.
PSM Executive Director Shabbir Imber Safdar is available to do interviews to explain the risks of this program to media. Contact him through editors@safemedicines.org and specify your print deadline.
Official actions and statements
- December 1, 2023: Texas Health and Human Services submits its Wholesale Prescription Drug Importation Program Report to the governor and state legislature.
Planning documents
- HB 25: Text | Fiscal Note | Bill Analysis
Background / resources
Just learning about the Texas foreign drug importation proposal? Start with some of these resources that outline the safety issues.
PSM Materials:
Op-eds from the Experts
Former DEA agent Doug Herber wrote this editorial, which was published on May 31, 2019 in the White Mountain Independent. In it, he writes that drug importation will cause “patients [to] unwittingly purchase foreign counterfeit drugs disguised as low-level medication, unaware of the dangers, end up as an overdose statistic. “
This editorial by Holly Strom and Kenneth Schell published in U.S. News and World Report on May 28, 2019 warns states considering drug importation that doing so will not keep costs down and also poses a safety risk to patients…
In this editorial, which was published in The Bend Bulletin on May 21, 2019, Canadian law enforcement veteran Don Bell explains why Oregonians can not rely on Canada for safe prescription drugs.
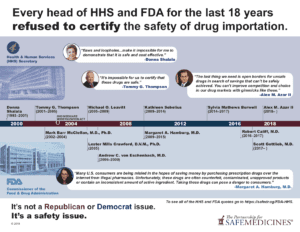
Dozens of the most highly qualified law enforcement officials and former, senior staff at the U.S. Food & Drug Administration have conducted in-depth analyses that show Canadian drug importation will lead to a massive increase in counterfeit drugs entering the U.S.
In this editorial published in The Hill on May 12, 2019, Brooklyn Roberts, the director of the health and human services task force at the American Legislative Exchange Council, discusses the risks of drug importation:
“The safety of our prescription drugs relies on a closed system where drugs can be traced to manufacturers, distributors, pharmacies and patients. Opening that system to foreign drugs would allow the potential for dangerous and potentially deadly medicines to land in the hands of the American public.”
In this editorial published on April 30, 2019, former FBI Director Louis Freeh talks about the safety risks of drug importation: “There are hidden risks and costs associated with the scheme that have not been getting much attention which impact your health and Colorado law enforcement’s ability to keep us safe.”
In this editorial published on April 26, 2019, Douglas Holtz-Eakin, economist and President of American Action Forum, questions economic truths about drug importation:
“Drug reimportation has long been the fool’s gold of health policy, and the Florida bill is no different. It flunks a basic policy analysis. But most amazing, it is drafted to raise hope, but not actually help Floridians.”
In this piece published in the Washington Free Beacon on April 25, 2019, staff writer Charles Fain Lehman explores issues around Florida’s drug importation proposal. “Critics,” he notes, “fear that the actual realities of regulatory oversight—especially in the hand of an as-yet-unnamed private vendor—will simply be too challenging to manage responsibly.”
In this piece, which was published in The Deseret News on April 24, 2019, pharmacist and Utah State Senate Majority Leader Evan Vickers raises serious concerns about importation as a strategy to lower drug prices:
“Anyone who truly understands how drugs are sold and distributed in the U.S. knows that there are very solid technical reasons that such importation is not viable. There are also serious concerns about drug safety, since the CHS cannot guarantee origin and purity on foreign-sourced drugs.”
In this piece, which was published on the ABC affiliate WJLA’s website on April 25, 2019, political analyst Boris Epshteyn explains that “this is a risky plan that will make it difficult to ensure that Floridians are receiving real and safe medicine.”