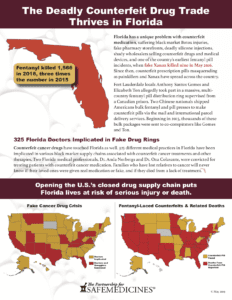
Florida House Bill 19 Would Expose Patients To Unregulated Prescription Drugs
It is no secret that many people across the U.S. have concerns about the cost of prescription drugs. These concerns have prompted legislators in multiple states to propose bills that would all drug importation. House Bill 19 (HB19) in Florida is an example of one of these bills currently under consideration. According to WFTV 9 in Orlando, HB19 would create two separate programs. The first program would make imported drugs available to Medicaid recipients, people who receive medical care at free clinics, inmates, and people treated at developmental disability centers and treatment facilities. The second program would allow the imported drugs to be sold directly to consumers at pharmacies within the state and faces additional federal hurdles before it can become law.
At a recent hearing in front of Florida’s Appropriations Committee, PSM’s executive director Shabbir Imber Safdar testified about the threat to patients posed to patients should the state circumvent the U.S.’s secure drug supply chain and, using history as an example, cautioned the legislators not to overestimate the savings this program might bring to the state. Other states tried drug importation in the past, and all ended in failure. After the hearing, Safdar said, “While PSM commends the Florida legislature for seeking ways to reduce out-of-pocket costs for their constituents, bringing prescription drugs into the state that came from outside of the U.S.’s secure drug supply chain is not the best or safest way to achieve this goal.”
For example, when Minnesota attempted drug importation starting in 2003, they envisioned as many as 700,000 residents participating in the program. At its height, the program filled only 1,100 prescriptions in a single month. Just before the program shut down in January 2010, only 57 prescriptions had been filled the previous month. In a letter sent to the then Governor Tim Pawlenty in 2004, the U.S. Food and Drug Administration (FDA) included a list of violations seen by the state’s own officials at preannounced visits which included:
- A pharmacy that had its pharmacists checking 100 new prescriptions or 300 refill prescriptions per hour, a volume so high that there is “no way to ensure safety”
- A pharmacy that failed to label its products even when sending multiple prescriptions in a single box
- Drugs that require refrigeration being shipped without any, meaning the drugs could lose stability and become less effective or not effective at all
- One pharmacy re-dispensed medications that were not labeled and had been returned by U.S. Customs and Border Protection
The letter also stated that Health Canada “cannot and will not guarantee the safety of drugs exported across the border through Internet pharmacies.”
In Florida, Representative Barbara Watson found a creative way to address any potential inequities over who would receive these drugs from questionable sources. Watson proposed several amendments to HB19 including Amendment 2. This amendment would have required that members of the Legislature, their staff, and their families who receive insurance through the state group health insurance program would also be required to use the drug imported through this program. The committee declined to adopt this amendment.
HB19 passed the Appropriations Committee and is now being analyzed by the Health and Human Services Committee. America needs to do something to rein in the cost of prescription drugs, but sacrificing the safety of our drug supply is not the answer.