August 25, 2025: New press draws attention to the dangers of poorly regulated medspas
Domestic News
Elle warned readers about medspas. An FDA inspection report revealed sterility concerts at an Indiana manufacturing facility.

Counterfeit Botox that sickened people across the country in April 2024 (FDA)
An August 2025 exposé in Elle about bad actors and loose regulation in the medical spa industry included the story of a Boston woman who falsely claimed to be a registered nurse and injected patients with counterfeit products from Brazil and China. Clients reported suffering from droopy eyelids and finding lumps of fake Botox or filler under their skin.
Weak regulatory control of the industry also came up when Josh Bolin of the National Association of Boards of Pharmacy appeared before the House Energy and Commerce in June. He cited a medical spa that had mailed customers syringes of GLP-1s from unregistered sources mixed and filled in a bedroom as an example of emerging threats to supply chain security.
On July 14, the Food and Drug Administration issued a form 483 after June and July inspections of an Indiana manufacturing plant owned by Catalant reporting that administrators’ “written records of investigations into the failure of a batch…do not always include the conclusions and follow-up,” notably failing to identify the root cause of foreign matter, including cat hair. Additionally, the facility provided insufficient instruction documentation for systems, sterility and quality control. According to Stat, similar problems turned up in 2022 and 2023 inspections. Novo Nordisk acquired the Catalant facility in late 2024.

FDA inspections like these are a cornerstone of U.S. medicine safety.
News about Prescription Drug Affordability Boards (PDABs)
On July 11, Colorado’s PDAB initiated rulemaking for an upper payment limit for Enbrel. At its July 15 meeting, Washington's PDAB continued to refine the data collection process for its work and chose Enbrel, Xtandi, Cabometyx, and Humira for affordability reviews. Oregon started reviews of Vraylar, Ajovy, Emgality, Nurtec, Ubrelvy and Entresto on July 16. On July 28, Maryland's board identified Farxiga and Jardiance as medicines with affordability challenges.
Legislation
Keep up with state legislation in the areas of pill presses, prescription drug affordability boards, and drug importation.
Patient safety issues in the GLP-1 space this week
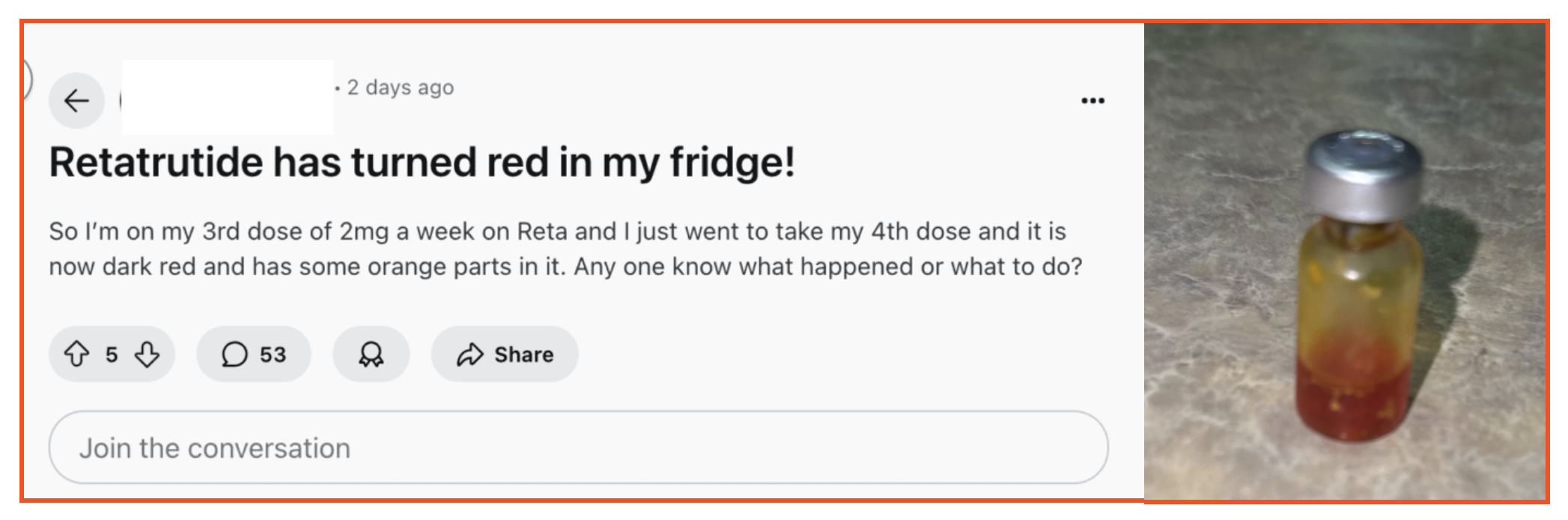
A user posted to Reddit on August 20th to ask what to do about their vial of purported retatrutide turning red and orange after having used three injections’ worth. Respondents speculated that the transformation was the result of mold or bad bacteriostatic water. They urged the poster to have the vial tested, which is a little bit like closing the barn door after the horses have escaped. Actual retatrutide is currently in phase 3 clinical trials, and is not available for patient use.
Reddit, August 20, 2025
The incident is a stark reminder that people taking versions of the as-yet unapproved weight loss medicine may not receive safe products and have no authority to turn to if anything unexpected happens.
International News
Diabetes Canada warned about fake products sold on social media. Taiwan drew praise for serializing exported pill presses.
The president of Diabetes Canada warned diabetes patients about a surge of fake diabetes products being sold online. In 2025, the organization has been contacted by at least 300 people who saw or bought fake diabetes products like smart watches claiming to track blood glucose levels or fake GLP-1 oral drops.
Officials at the Cross-Border Drug Enforcement Cooperation Forum, hosted by the U.S. Drug Enforcement Administration, praised Taiwan for increasing regulation of pill presses by serializing exported pill press machinery.
The U.K.’s Medicines and Healthcare products Regulatory Agency advised parents to stop using Nutrition Ignition Kids Magnesium Glycinate Gummies, because it contains undeclared melatonin, which is prescription-only in the U.K..
Authorities in Bangladesh fined a pharmaceutical company for selling a counterfeit “anti-itch drug” falsely branded as an imported product from the Philippines.