March 30, 2026: A second pharmacy benefit manager settles suit with the FTC over insulin price inflation
Major Stories
The Federal Trade Commission (FTC) has reached a settlement with CVS Health’s Caremark in its insulin pricing lawsuit. In September 2024, the FTC brought cases against the “Big Three” pharmacy benefit managers, Caremark, Express Scripts, and Optim RX, alleging they inflated insulin prices by engaging in anticompetitive practices. Last month, Express Scripts settled its case with the FTC, agreeing to several business changes that would address the cost of insulin. According to Reuters, the Caremark settlement is similar to the agreement the FTC reached with Express Scripts.
Domestic
The FDA clarified that is isn't permissible for alternative funding programs to import medication. The Partnership for Safe Medicines was cited in a House subcommittee hearing about FDA's destruction authority. Six people were arrested in connection with operations that involved pill presses.
Buying from pharmacy-to-pharmacy marketplaces? Our Suspicious Sales Surveillance Report reveals just how often “too good to be true” deals may signal diverted or counterfeit medicines that put patients at risk. Learn what red flags to watch for, and how to protect your pharmacy and stay compliant, before making your next purchase.
The U.S. Food and Drug Administration (FDA) responded to a citizen petition from Aimed Alliance, clarifying that alternative funding program (AFP) facilitation of prescription drug importation is not permissible under federal law. The agency also emphasized that the cost of a drug is not a factor in its case-by-case decisions around personal importation and reinforced the need for clear reporting pathways for potentially unauthorized practices. Read the materials that we received through open record requests to towns and school districts that use AFP services, including invoices for imported medications.

Items seized in the Los Angeles raid (Photo: DEA)
A 2025 letter from the Partnership for Safe Medicines to Congress was cited by Representative Troy Carter in the Health Subcommittee of the House Committee on Energy and Commerce. The hearing covered HR 2715, the Destruction of Hazardous Imports Act, which would allow the FDA to destroy products that it believes pose a public health risk. Our letter, which was cosigned by multiple patient safety organizations, expressed support for the FDA’s fiscal year 2026 proposal to grant it destruction authority.
The Los Angeles Homeland Security Task Force announced the seizure of 500,000 counterfeit M30s, Xanax, oxycodone & ecstasy pills and 15 kilograms of fentanyl in a major bust.
Law enforcement arrested six individuals in the Bronx, New York after discovering a fentanyl and heroin packaging operation inside an apartment, seizing over six pounds of fentanyl valued at more than $500,000. Investigators also uncovered counterfeit “xylazine” bottles that actually contained other substances, along with large quantities of drug packaging materials spread throughout the residence.
Pill Presses
The U.S. Drug Enforcement Administration (DEA) announced that Phase II of Operation Fentanyl Free America, which ran from January 10 to February 12, 2026, included the seizure of 29 pill press machines. 55 machines have been seized since the operation was launched in October 2025.
Huntsville, Alabama, police arrested four people after the search of a home yielded a pill press, 1.3lbs of opiates, and 24lbs of etizolam in a home. In Florida, two men were arrested after authorities seized a pill press at a motel.

The DEA shared this photo of one of the pill presses they seized
Regulatory action
The FDA issued a warning letter to a Chinese manufacturer of over-the-counter pharmaceuticals for violations of Current Good Manufacturing Practice (CGMP) regulations. The firm failed to conduct testing of incoming components, including active pharmaceutical ingredients, relying on the suppliers' testing without verifying the quality of the products. Additionally, they did not adequately batch test finished products to confirm the identity and strength of active ingredients.
Legislation
Virginia HB483 and SB271, the state's bills to establish a PDAB with the ability to set an upper payment limit, have passed the House and Senate and are awaiting the governor's signature.
Keep up with state legislation in the areas of pill presses, prescription drug affordability boards, drug importation, and med spas.
International
Regulators and advocates in Canada, Nigeria, Pakistan, and Vietnam warned the public about counterfeit drugs sold everywhere from a massage parlor to social media.
Vietnamese health authorities warned about counterfeit, unlicensed HIV prevention injections (YEZTUGO/lenacapavir) being sold on social media, allegedly by a former nurse falsely claiming affiliation with a children’s hospital. Officials confirmed the drug is not approved for use in Vietnam and urged the public to avoid it due to serious safety and authenticity concerns.
Health Canada warned the public about unauthorized and counterfeit products at a Mississauga massage parlor, including counterfeit tirzepatide products, including Mounjaro vials and KwikPens with invalid batch numbers, hyaluronic acid hair care products, and Biacto. Health Canada advised consumers to buy drugs from a licensed pharmacy with a valid prescription to ensure the drugs are safe.
Pakistan's shortage of cancer medications has caused a sharp rise in counterfeit products entering the local market. Oncologists are warning that patients may be exposed to ineffective or dangerous treatments by seeking recommended drugs through the unregulated supply chain. One Karachi oncologist, whose patient purchased pembrolizumab (Keytruda) for a few thousand rupees, told The News Pakistan, “even the vial did not look original. The solution was not clear, there were visible particles, and the quantity appeared less than the standard presentation.”
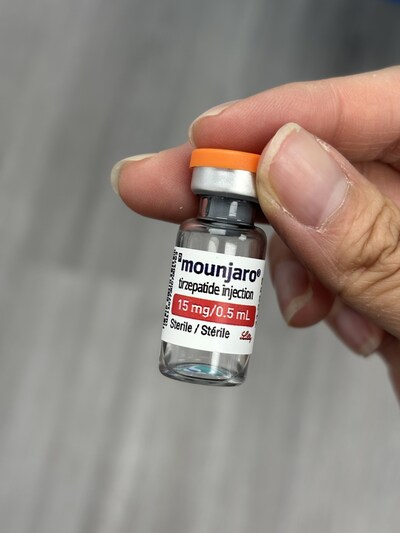
(Photo: CNW Group/Health Canada (HC))
Nigerian regulators alerted the public to the circulation of a batch of counterfeit Herceptin 600 mg. The drug, which is used to treat stomach cancer, has the batch number A8519B34 and was reported in Lagos State.

