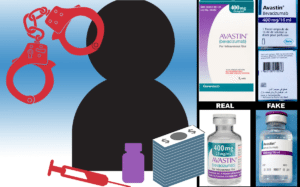
Narinder Kaulder, head of Operations for River East, a company named in the Canada Drugs indictment, is currently fighting extradition from England on charges he was part of the conspiracy that sold counterfeit cancer medication to U.S. oncology clinics in 2012, the Daily Mail reports. According to the CanadaDrugs Indictment, lot number B6011 of Avastin that…
The Morning Consult recently published an op-ed by Libby Baney, the Executive Director of the Alliance for Safe Online Pharmacies (ASOP Global), a nonprofit dedicated to addressing the growing public health threat of illegal online pharmacies. In the piece, Ms. Baney stated that drug importation proposals are not a safe or effective way to address the rising cost of prescription drugs . . .
Ronald Piervincenzi, CEO of the U.S. Pharmacopeial Convention describes the dangerous flaws in any plan to open up U.S. borders to wholesale prescription medication importation from Canada.
The Washington Examiner recently ran an op-ed by Charlie Cichon, the Executive Director of the National Association of Drug Diversion Investigators. In it he explained how the opioid crisis will only be made worse if the U.S. allows drug importation.
The Newfoundland and Labrador Pharmacy Board (NLPB), which governs and regulates all pharmacies operating in their Canadian province, has written a letter to the U.S. Congress, which bluntly states, “the importation of medicines from Canada is not the solution.”
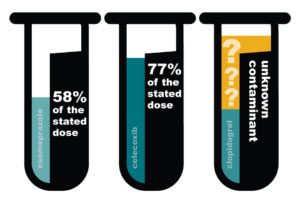
Canadian pharmaceutical importer Quantum Solutions and its officers have been charged with two felony counts by the DOJ. Quantum Solutions runs a dozen different pharmacy websites, including Canada Drug Center. Dr. Kenneth McCall, of the University of New England, tested prescriptions from Canada Drug Center in 2014, and found they were substandard and contaminated. Three…
The College of Pharmacists of Manitoba (CPM), the regulatory authority in the Canadian province of Manitoba that oversees safe pharmacy practice, wrote to the United States Congress requesting that they reject the current importation bill.
University of Maryland professor Robert Freeman warns that proposed changes in U.S. laws and regulations to make the movement of drugs from other countries easier will increase fentanyl deaths in the U.S. In an editorial in The Hill, Dr. Freeman warns that the opioid abuse problem affects a greater proportion of the Canadian population…
Former Federal Regulators, Law Enforcement Officials Dispute Rhetoric on Drug Importation Merits, Highlight the Serious Harm Posed to Patient Health Washington, D.C. — In one of the strongest challenges to drug importation proposals to date, several former federal law enforcement officials and public health experts warned of serious consequences in opening up the U.S. drug supply…
A small-scale study in the United Kingdom has found that 75% of the assessed online pharmacies that offer drugs for sale in the UK are unregistered as legitimate pharmacies, and most offer antibiotics without a prescription. This finding could be a serious driver of microbial resistance. Obtaining Antibiotics Online from within the UK: a Cross-Sectional…