CNN Warns that Fake Drugs are a Global Industry That Puts Your Life at Risk
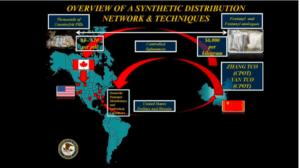
Source: U.S. Department of Justice
In a Mosaic Science article shared by CNN, author Srinath Perur delves into the global pharmaceutical industry and discovers that in India, counterfeit and substandard medications are an everyday occurrence.
In 2012 60 people in Pakistan died from cough syrup that was contaminated with an incredibly strong opioid called Levomethorphan. The same medication found its way to Paraguay, and sickened 44 in 2013. By the time the World Health Organization sent out a global alert concerning the contaminated cough syrup ingredient, batches of the medication from this same Indian factory had made their way to multiple countries in Europe, north Africa, the Middle East and Latin America. The widespread distribution of this toxic medication was finally stopped, but only after traveling and poisoning around the globe.
Outside the United States, the most pressing counterfeit drug concern is counterfeit and substandard antimicrobials and antibiotics that breed drug resistance. In the article, Perur reports Indian counterfeit drug investigator Suresh Sati describing the ease with which one can purchase substandard medications for resale and the large quantity of counterfeit samples he had collected. “Within 15 minutes of sitting down, he has produced so many samples of fake medicines that our tumblers of coffee risk being crowded off the table. He collected them over years of investigations, from raids or from informants in fake medicine distribution networks.”
CNN reports that a commonly prescribed antibiotic sold in India called Zifi is available in counterfeit versions in a blister pack that are indistinguishable from the genuine drugs. Sati also explained how easy it was to get illicitly manufactured medications of varying quality, depending on how much one was willing to pay. Sati claimed manufacturers would ask; "Which do you want? Full-salt, half-salt or chalk-mitti?" meaning “full-active ingredient, half-active ingredient, or only chalk.”
According to a report from an April article in The International News, “The Drug Testing Laboratory of Punjab has undertaken rigorous testing of medicines available at primary, secondary and tertiary care hospitals. The results are shocking. Most of the medicines tested are unfit for human consumption. The number is as high as 80 percent of all medicines tested.”
Counterfeit medications are not as widespread in the US, but they are available. In 2018, the United State Food and Drug Administration Office of Criminal Investigations prosecuted a number of counterfeit medication sellers in this U.S. In cases where counterfeit drug sellers are working within U.S. borders, fake drugs criminals are easy to prosecute.
However, when such clandestine medication brokers operate in other countries such as Canada and China, it is harder to bring them to justice in a meaningful way. The CanadaDrugs.com case and the current indictment of the Zheng organization in Shanghai illustrates how challenging it is to impose U.S. laws on criminals operating outside U.S. borders.