Posts Tagged ‘security-page-CBP’
Prescription Drug Freight Fraud Report, April 2026
What do FDA records of GLP-1 and antibiotic imports show about freight fraud patterns over the last six months?
[...]What’s master carton smuggling?
In March, customs officers in Cincinnati announced the seizure of thousands of shipments of GLP-1s and other peptides packaged inside more than 300 “master cartons” with deceptive manifests meant to slip into the country unnoticed. Curious how this smuggling scheme works? Read on.
[...]Prescription Drug Freight Fraud Report, January 2026
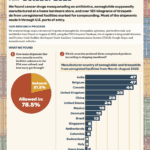
The last two months of 2025 saw thousands of shipments of drugs popularized by social media imported into the U.S., as well as Xarelto and antibiotics. The shipments carried a wide range of product codes; some appropriate, and others far less so.
[...]Prescription Drug Freight Fraud Report, November 2025
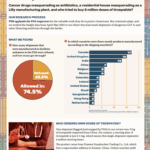
Our analysis of data in September and October 2025 revealed that large-scale freight fraud involving GLP-1s, oncology drugs, and other high-demand pharmaceuticals continues unabated.
[...]Prescription Drug Freight Fraud Report, October 2025
What large-scale commercial imports made it into the country between March and August of 2025?
[...]Prescription Drug Freight Fraud Report, September 2025
Our analysis of data from June and July revealed hundreds of pharmaceutical shipments that entered the U.S. from facilities no one would expect, including unregistered Chinese exporters and alternative medicine clinics abroad.
[...]PSM leads letter in support of an FDA mandate to destroy high-risk imports
PSM led a coalition of organizations urging leaders of the Senate HELP and House Energy and Commerce committees to help strengthen the FDA’s ability to protect Americans from unsafe and counterfeit medicines and medical products.
[...]Prescription Drug Freight Fraud Report, July 2025
What if your tirzepatide shipment came from a Brazilian beauty clinic? Or a vial of semaglutide was manufactured, supposedly, at a Costco in Toronto? In April and May 2025, dozens of shipments of semaglutide, tirzepatide, apixaban, and antibiotics entered the U.S. from facilities that aren’t in the FDA’s drug manufacturing database. These aren’t low-volume personal-use…
[...]Prescription Drug Freight Fraud Report, May 2025
What if your blood thinners were made in an unverified location in Colombia? In March 2025, dozens of pharmaceutical shipments entered the U.S. that were manufactured in places no one would expect.
[...]PSM submits comment on CBP’s proposed rules to improve screening of low-value shipments
On March 14, the Partnership for Safe Medicines submitted comment on Customs and Border Protection’s proposed update to regulations around low-value, “de minimus” shipments.
[...]