Article Topic
Case overview: Gilead Sciences v Pain Relief Rx, et al (2026 — ) – ongoing
Gilead Sciences has filed a complaint against Pain Relief Rx and other companies that it alleges sold diverted medicines on pharmacy to pharmacy marketplaces.
[...]Partnership for Safe Medicines Applauds FDA Action to Curb Unsafe Compounding of GLP-1 Medications
The Partnership for Safe Medicines strongly supports the Food and Drug Administration’s proposal to exclude semaglutide, tirzepatide, and liraglutide from the 503B bulks list. This action reflects sound science, sound law, and a clear-eyed commitment to patient safety. Full statement inside.
[...]Prescription Drug Affordability Board Activity, February-March 2026
Prescription Drug Affordability Board Activity, February and March 2026 Activities Summary Colorado: The PDAB did not meet in February or March. The next meeting was held April 3, 2026. Maryland: At its February meeting, the PDAB introduced a new board member and approved UPL frameworks for Ozempic and Trulicity. In March, it heard a briefing…
[...]Jimmie Wilson suffered acute liver failure after taking compounded weight loss shots
Jimmie Wilson suffered liver failure after being treated with compounded tirzepatide. She didn’t know she was taking any risks, or that compounded and name-brand drugs weren’t the same.
[...]The FDA speaks up about AFPs and drug importation
Is personal drug importation legal in the context of alternative funding programs? The FDA says no.
Are self-funded health plans allowed to import medicine because of “Section 804” or “Florida’s importation plan”? Also, no.
[...]Case overview: USA v Adam Brosius, Patrick Boyd and Charles Boyd (2024 – 2026)
Adam Brosius and Charles and Patrick Boyd were proprietors of Safe Chain Solutions, a Maryland drug wholesaler that sold distributed misbranded and adulterated drugs and trafficked medicine products with false documentation. The men have been sentenced to a combined 46 years in prison.
[...]Photo gallery, USA v Boyd exhibits
Photo gallery, USA v Boyd exhibits Two bottles of Biktarvy, lot CCZCBA. (Exhibit 30) A Biktarvy bottle filled with the antipsychotic Seroquel. (Exhibit 192) A Biktarvy bottle filled with the antipsychotic Seroquel. (Exhibit 192) A Biktarvy bottle filled with the antipsychotic Seroquel. (Exhibit 192) Seroquel found in a Biktarvy bottle. (Exhibit 192) Patient’s photo of…
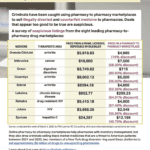
[...]Using pharmacy-to-pharmacy marketplaces? Read this Suspicious Sales Surveillance Report
Online pharmacy-to-pharmacy marketplaces help with inventory management, but they also require vigilance because they carry a higher risk of being exploited by criminals selling black market medicines.
[...]Counterfeit drug crimes typically earn light sentences. How did Safe Chain’s co-owners, the Boyd brothers, end up with 18 and 20 years?
Wire fraud charges drove much higher penalties than the shipping of unsafe medicine into the supply chain, and the Boyds’ sentences were based on the government proving high-dollar, deliberate fraud.
[...]Plea Agreement, USA v Sanjay Kumar
United States District Court Southern District of Texas, Houston Division USA v Sanjay Kumar Plea agreement Filed October 2025 Read the document.
[...]