Industry experts told us that these two companies are on the FDA’s Green List, despite problematic inspections in late 2024 and early 2025. Learn what the inspectors found.
Well-meaning advocates continue to propose importing medicine from foreign countries to lower drug prices for Americans. Past efforts didn’t pay off financially and raised public health risks. Here’s the state of this issue today.
A Pennsylvania compounding pharmacy received another FDA warning letter following a series of enforcement actions, including a $1 million state fine in October.
The last two months of 2025 saw thousands of shipments of drugs popularized by social media imported into the U.S., as well as Xarelto and antibiotics. The shipments carried a wide range of product codes; some appropriate, and others far less so.
On February 17, PSM submitted written and oral testimony in opposition to Colorado HB26-1056, which would permit self-insured employers to use alternative funding programs to import foreign medicines from outside the U.S. drug supply chain.
Unregulated pill presses and molds make fake pills with dangerous and even deadly consequences for thousands of people every year. Read our July 2025 to January 2026 update about pill press seizures, policy developments, and legislation.
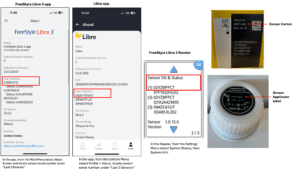
The FDA has posted a notice about the recall of continuous glucose monitors from Abbott.
The federal spending package that ended January’s government shutdown will prohibit PBMs from linking rebates to drug companies’ list prices. The FTC reported that PBMs inflated the prices of generic drugs.
The FDA has posted a notice for a dietary supplement recalls involving products found to contain undeclared pharmaceutical ingredients.
PSM Executive Director Shabbir Imber Safdar released the following statement in response to news that Hims & Hers is now compounding pill versions of a newly approved GLP-1 weight-loss drug.